  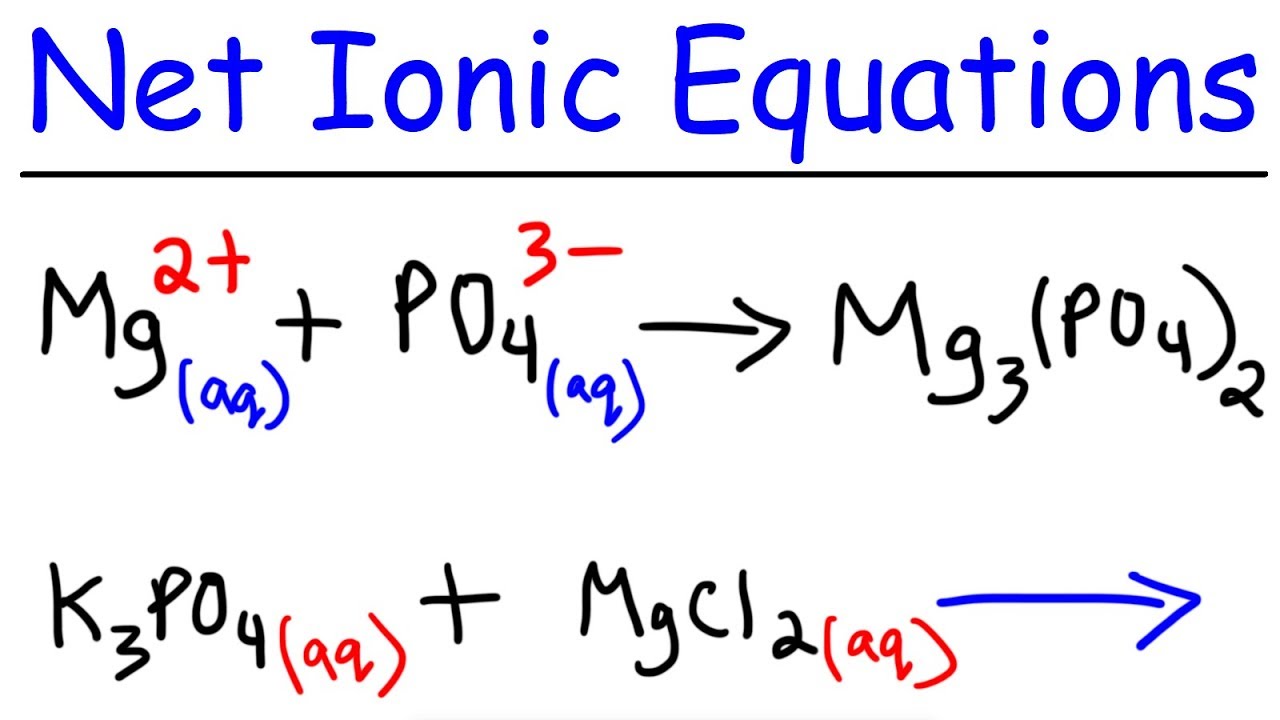
Watch these demonstrations of how to test for. For example, when a solution of silver nitrate is added to a solution of. (You cannot swap both you would end up with the same substances you started with.) Either perspective should allow you to predict the proper products, as long as you pair a cation with an anion and not a cation with a cation or an anion with an anion. 1 2 3 4 5 6 7 8 Gas tests When a reaction produces a colourless gas, it is straightforward to test if this is hydrogen, oxygen or carbon dioxide. Precipitate or the deposit is an insoluble solid formed by reactions in a solution. When the RSS is small, precipitation is more likely to occur by particle growth than by nucleation. A solution with a large, positive value of RSS has a high rate of nucleation and produces a precipitate with many small particles. A basic definition of an ionic compound is that they are molecules that consist. 1 Analogously, in medicine, coprecipitation is specifically the precipitation of an unbound 'antigen along with an antigen-antibody complex'. with n(H 2) on the vertical axis and percentChemistry is defined as the systematic. The numerator of equation 8.2.12, Q S, is a measure of the solute’s supersaturation. In a precipitation reaction two soluble ionic compounds react to give an. In chemistry, coprecipitation ( CPT) or co-precipitation is the carrying down by a precipitate of substances normally soluble under the conditions employed. 
There are two equivalent ways of considering a double-replacement equation: either the cations are swapped, or the anions are swapped. Chemistry 101 Experiment 7 - ENTHALPY OF REACTION USING HESSS LAW. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
